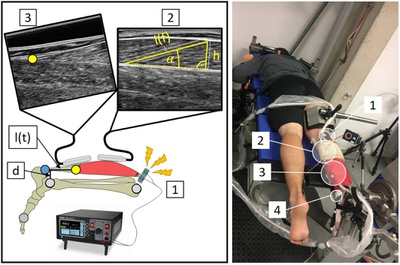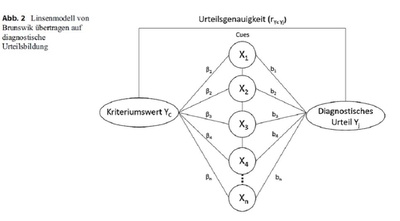
Commentary: Do all studies using medical devices fall under the European Medical Device Regulation?
24 September 2024
Rethwilm, R., Schecklmann, M., Agboada, D., Ole Bergmann, T., & Seiberl, W. (2024). Commentary: Do all studies using medical devices fall under the European Medical Device Regulation? Clinical Neurophysiology. https://doi.org/10.1016/j.clinph.2024.09.012
Commentary on: A. Antal et al. (2024). The consequences of the new European reclassification of non-invasive brain stimulation devices and the medical device regulations pose an existential threat to research and treatment: An invited opinion paper. Clinical Neurophysiology. https://doi.org/10.1016/j.clinph.2024.03.039
“We will argue that most research studies using medical devices (MDs) do not fall under the MDR, rendering the considerable administrative and financial burdens associated with ‘clinical investigations’ obsolete.”
Funding
This project was funded by partly funded by the dtec.bw – Digitalization and Technology Research Center of the Bundeswehr (MEXT project). dtec.bw is funded by the European Union – NextGenerationE.
Correspondence:
Professur für Bewegungswissenschaften mit einem Schwerpunkt Digitalisierung